
Carbon monoxide is poisonous to all warm-blooded animals (when it is inhaled and combined with hemoglobin in the blood, which prevents the absorption of oxygen) and to many other life forms. It is a colorless, odorless, tasteless, toxic gas. CO 3).Carbon monoxide is a compound of carbon and oxygen with the chemical formula CO. Step 6 - Brackets may be used around radicals (groups of atoms that are charged e.g.Na 1 Cl 1 or Mg 2 O 2), ignore them and rewrite the formulae without them (e.g. Step 5 - If the numbers on each part are the same (e.g.Step 4 - Write the completed formulae with those same numbers at the bottom.Cross-over the top valency number to the bottom of the other ion symbol. Step 3 - Now ignore the positive and negative signs.Step 2 - Using the valency table, write the two ions and their valencies.Na 1+) and the second is a negative ion (e.g. Step 1 - In the ionic compounds to be learnt in junior science, there are two parts to the ionic compound - the first is a positive ion (usually a metal e.g.For example in the ionic compound sodium chloride NaCl, the chlorine ion (Cl 1–) gains one electron that was given by the sodium ion (Na 1+).Ĭommon Elements and Symbols to Learn Element Symbol Ionic Compound - a compound formed from the attraction between positive and negative ions.Covalent Compound- a compound where electrons are shared between the atoms (e.g.In chemical reactions, atoms tend to gain or lose electrons to resemble the electron numbers of the stable Noble Gases.Negative Ions are atoms that have gained electrons (e.g.Positive Ions are atoms that have lost electrons (e.g.An atom gaining or losing electrons will get an overall charge. When atoms react, they may either gain or lose electrons.The electron rings of these unreactive gases are full, therefore they become stable.Inert or Noble Gases are unreactive gases.Molecule - A molecule is the smallest particle of either an element or a compound.Compound - A compound is a substance made of more than one type of atom (e.g.Element - An element is a substance composed of the same type of atoms (e.g.Electron rings close to the nucleus are filled first.A limited number of electrons are situated in each electron 'ring'.  
Electrons orbit the nucleus in electron levels or 'rings'. 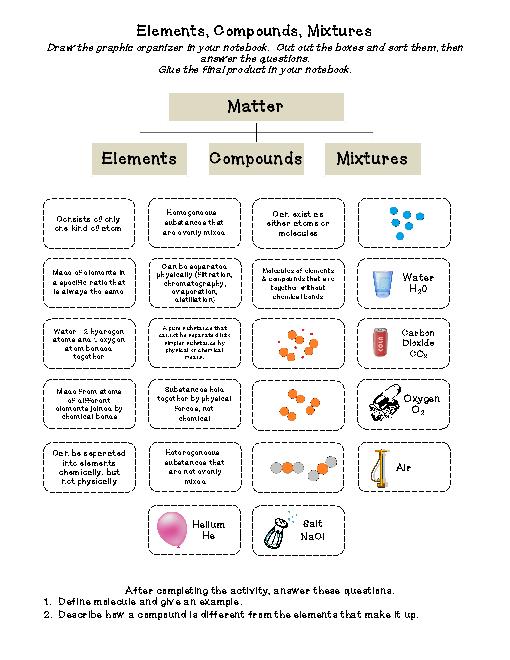
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
